Keep Your Eyes Open to Plant Diseases Newsletter
A newsletter produced at the Texas Plant Disease Diagnostic Lab by the Diagnostician (S. McBride).
Keep your eyes open Vol.1 No.1
Keep your eyes open Vol.2 No.1
Archived News from the old website
June 2010
Powdery mildews
 As temperature rises in all parts of Texas, the environment may still be humid from the spring rains. This is the time when the plant disease, POWDERY MILDEW, likes to appear. Powdery mildew is the common name of a disease that is “catch-all” for several fungi that have similar characteristics and can be managed in a similar fashion. Many plants are affected by powdery mildew include landscape ornamentals such as crape myrtles, roses and zinnia; and vegetables such as cucumbers, melons and peppers. In all cases, the powdery mildew appears as a dusty white to off-white coating on the affected plant part – typically leaves. It starts as tiny discrete round white spot. As it grows, these spots can coalesce and form large “blotches” of white powder mats. The spores of these fungi can be observed under the microscope and are used to confirm the diagnosis. Common damage caused by these fungi are stunted and distorted leaves or buds, sometimes resulting in premature defoliation. On vegetable, this disease can severely impact yields.
As temperature rises in all parts of Texas, the environment may still be humid from the spring rains. This is the time when the plant disease, POWDERY MILDEW, likes to appear. Powdery mildew is the common name of a disease that is “catch-all” for several fungi that have similar characteristics and can be managed in a similar fashion. Many plants are affected by powdery mildew include landscape ornamentals such as crape myrtles, roses and zinnia; and vegetables such as cucumbers, melons and peppers. In all cases, the powdery mildew appears as a dusty white to off-white coating on the affected plant part – typically leaves. It starts as tiny discrete round white spot. As it grows, these spots can coalesce and form large “blotches” of white powder mats. The spores of these fungi can be observed under the microscope and are used to confirm the diagnosis. Common damage caused by these fungi are stunted and distorted leaves or buds, sometimes resulting in premature defoliation. On vegetable, this disease can severely impact yields.
April 2010
Springing up – problems on turf
 Spring time – diseases are also springing up this time of the year. In Texas, a couple of “patch” problems are common in our warm season grasses. Look out for the smoky halo at the edge of a bare patch that is characteristic of “brown patch” caused by the fungusRhizoctonia sp. This disease develops quickly when air temperatures are between 75° and 85° F AND conditions are wet.(ie. leaves are wet for prolonged periods). Hence some simple non-chemical activities can help to prevent this problem. Action taken to improve soil drainage, reducing shade, proper irrigation practices and/or improving air movement will help with reducing residual moisture on the leaves. Many lawn fungicides available to homeowners are active against Rhizoctonia. Please read labels carefully for proper usage and rates. Unfortunately, this fungus is difficult to destroy once it is established the turfgrass. Also, this is not the only problem that you might encounter in your lawn.
Spring time – diseases are also springing up this time of the year. In Texas, a couple of “patch” problems are common in our warm season grasses. Look out for the smoky halo at the edge of a bare patch that is characteristic of “brown patch” caused by the fungusRhizoctonia sp. This disease develops quickly when air temperatures are between 75° and 85° F AND conditions are wet.(ie. leaves are wet for prolonged periods). Hence some simple non-chemical activities can help to prevent this problem. Action taken to improve soil drainage, reducing shade, proper irrigation practices and/or improving air movement will help with reducing residual moisture on the leaves. Many lawn fungicides available to homeowners are active against Rhizoctonia. Please read labels carefully for proper usage and rates. Unfortunately, this fungus is difficult to destroy once it is established the turfgrass. Also, this is not the only problem that you might encounter in your lawn.
March 2010
Plant Disease and the compost pile
 A question that I get quite a bit is “Can I compost diseased plant material?” My answer has always been a question – “How confident are you in your composting ability to completely heat the pile?” The short end of this story – it is difficult to ensure that every part of the disease plant material gets fully composted. Also, not all pathogen are alike. Although many plant pathogens would be killed/inactivated by the heat from the compost pile, some might just survive and are capable of causing new infections. Nonetheless, there is still benefit to composting. For example, tomato plant with early blight infection (caused by fungus Alternaria solani) is a common problem to many backyard growers in Texas and beyond. Through the composting process, if done properly, much of the fungus would be killed. What about the escapee? The lower number (if present) may be held in check by other microbes that are in the finished compost. There is always the possibility that conducive conditions might occur in the next season for fungus to develop diseases.
A question that I get quite a bit is “Can I compost diseased plant material?” My answer has always been a question – “How confident are you in your composting ability to completely heat the pile?” The short end of this story – it is difficult to ensure that every part of the disease plant material gets fully composted. Also, not all pathogen are alike. Although many plant pathogens would be killed/inactivated by the heat from the compost pile, some might just survive and are capable of causing new infections. Nonetheless, there is still benefit to composting. For example, tomato plant with early blight infection (caused by fungus Alternaria solani) is a common problem to many backyard growers in Texas and beyond. Through the composting process, if done properly, much of the fungus would be killed. What about the escapee? The lower number (if present) may be held in check by other microbes that are in the finished compost. There is always the possibility that conducive conditions might occur in the next season for fungus to develop diseases.
If you have to find out what to do with your disease plant material, I would suggest that you learn more about the disease(s) that has occurred on the plant. Understanding more about the pathogen will give some insight as to whether that disease plant material might be okay for composting. There is always some risk. If you do not like risk or potential unknown or do not wish to do homework on the pathogen(s), then I would suggest to stay away from adding any disease plant material into your compost pile.
For those who might be interested, a recent paper in the Journal “Crop Protection” reported results from a study from Spain that showed composting of disease melon and pepper plant reduced the viability of a couple of viruses (Pepper mild mottle virusand Melon necrotic spot virus) and a soilborne fungus(Olpidium bornovanus). [ Aquilar et al. Crop Protection 29 (2010) 342-348.]
To learn more about composting, visit the Master Composting site (http://mastercomposter.com). AgriLife Extension at one time put together a resource for kids – “Composting for kid” – click to check this resource.
February 2010
Time to get the beds ready: Compost as plant disease detterent.
January 2010
Into the deep freeze
 In the first full week of the New Year, many parts of Texas is expected to experience unusually cold weather. Very cold conditions are not nice to many plants. I expect that we will have some reports of freeze damage on plants succeeding this cold weather anomaly. Some plants are capable of withstanding the cold naturally. However, many of the plants that we grow in our yards are susceptible to freeze damage. (Photo: Minor freeze damage -dead tips, on exposed manihot plant)
In the first full week of the New Year, many parts of Texas is expected to experience unusually cold weather. Very cold conditions are not nice to many plants. I expect that we will have some reports of freeze damage on plants succeeding this cold weather anomaly. Some plants are capable of withstanding the cold naturally. However, many of the plants that we grow in our yards are susceptible to freeze damage. (Photo: Minor freeze damage -dead tips, on exposed manihot plant)
A few things to know:
- If a plant is still actively growing (not dormant), it is susceptible to freeze damage – particularly the actively growing part.
- Containerized plants are susceptible to freeze damage as soil can freeze from all sides of the pot.
- Plants with poorly established roots and that are stressed are more susceptible to freeze damage.
- Lack of soil moisture can make a freeze more damaging.
So what can we do to protect our susceptible plants?
- Bring containerized plants indoors or into a protected area out from cold (ie. garage or shed.)
- In areas where there has been lack of rain, irrigate plants 24 to 48 hours prior to freeze incidence.
- Cover cold sensitive plants: one can use blankets, layers of newspapers, insulation layers or crop covers(remember to weigh sides down to prevent cover material from being blown off).
- Protect roots that are shallow or on new plants (not yet established). Use methods such as listed in #3
1/15/2010 Update: Looking around town in Brazos Valley, I have not seen too much immediate damage from the freeze. I suspect plants that are in open areas would tend to be affected more. Continue to keep an eye on trees and turf to see if there if any after effects when green up begins in a few weeks.
November/December 2009
What’s in your water?
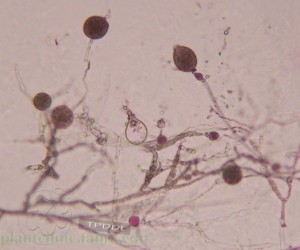 Two most common plant pathogen disseminated through contaminated irrigation waters are Pythium and Phytophthora. Pythium is mainly a problem during propagation and growth of herbaceous material. Phytophthora is an important pathogen on a number of ornamental crops. It can cause root rot, cankers, and foliar blights. Cankers and foliar blights are readily diagnosed since the symptoms are apparent. Root rots are more difficult to diagnosed since the roots are obscured from view. Early symptoms of stunting may not be readily apparent in a block of plants that are uniformly infested. Severe symptoms include mimics of nutrient deficiency damage symptoms, and eventual plant death. Phytophthora is easily spread through water and can be present in irrigation water when irrigation runoff is recycled. Ponds, irrigation water, and soil can be tested for the presence of Phytophthora. Tests for the presence of Pythium are misleading since there are a number of species that are found in irrigation water but do not cause important diseases. Anyone who irrigates with surface water is potentially at risk of contamination from Phytophthora. Facilities that catch runoff and reuse it for irrigation are especially at risk. A large number of ornamental plants are particularly susceptible to diseases caused by various species of Phytophthora. Producers of these crops should consider testing for Phytophthora. If diseased plants have been diagnosed with Phytophthora, testing may help determine the source and distribution of Phytophthora. Producers that use well water or municipal water sources do not need to have water tested. Pond sampling is the most sensitive method but requires access to the pump intake in the pond. Irrigation water sampling is not as sensitive but more convenient. Soil are tested using a baiting technique to recover the Phytophthora. (by Karl Steddom)
Two most common plant pathogen disseminated through contaminated irrigation waters are Pythium and Phytophthora. Pythium is mainly a problem during propagation and growth of herbaceous material. Phytophthora is an important pathogen on a number of ornamental crops. It can cause root rot, cankers, and foliar blights. Cankers and foliar blights are readily diagnosed since the symptoms are apparent. Root rots are more difficult to diagnosed since the roots are obscured from view. Early symptoms of stunting may not be readily apparent in a block of plants that are uniformly infested. Severe symptoms include mimics of nutrient deficiency damage symptoms, and eventual plant death. Phytophthora is easily spread through water and can be present in irrigation water when irrigation runoff is recycled. Ponds, irrigation water, and soil can be tested for the presence of Phytophthora. Tests for the presence of Pythium are misleading since there are a number of species that are found in irrigation water but do not cause important diseases. Anyone who irrigates with surface water is potentially at risk of contamination from Phytophthora. Facilities that catch runoff and reuse it for irrigation are especially at risk. A large number of ornamental plants are particularly susceptible to diseases caused by various species of Phytophthora. Producers of these crops should consider testing for Phytophthora. If diseased plants have been diagnosed with Phytophthora, testing may help determine the source and distribution of Phytophthora. Producers that use well water or municipal water sources do not need to have water tested. Pond sampling is the most sensitive method but requires access to the pump intake in the pond. Irrigation water sampling is not as sensitive but more convenient. Soil are tested using a baiting technique to recover the Phytophthora. (by Karl Steddom)
August 2009
The TPDDL and Pierce’s Disease Diagnostics
 The TPDDL has now developed the capacity to diagnose Pierce’s Disease (PD) of grapes by applying the most progressive and reliable laboratory methods. These methods have been tested in a number of projects sponsored by the Texas Pierce’s Disease Research and Education Program and have been successfully applied in those projects to confirm PD throughout the State. Growers will have the option to select the protocol they prefer, and will be expected to fully participate in the diagnostic process by filling out the proper forms and following some simple sampling directions. This system has been developed to provide grape growers with the best possible diagnostic results.
The TPDDL has now developed the capacity to diagnose Pierce’s Disease (PD) of grapes by applying the most progressive and reliable laboratory methods. These methods have been tested in a number of projects sponsored by the Texas Pierce’s Disease Research and Education Program and have been successfully applied in those projects to confirm PD throughout the State. Growers will have the option to select the protocol they prefer, and will be expected to fully participate in the diagnostic process by filling out the proper forms and following some simple sampling directions. This system has been developed to provide grape growers with the best possible diagnostic results.
The diagnosis of Pierce’s disease can be a difficult problem. The bacterium, Xylella fastidiosa, may be unevenly distributed in host vines and typical symptoms may not be an accurate measure of the degree of colonization by the pathogen. Therefore, it is important that samples be collected by following the instructions on the Pierce’s Disease Diagnosis Form (TPDDLform1004). It will be noted that two laboratory assay protocols are available. They are ELISA (Enzyme Linked Immunosorbent Assay) and QRT-PCR (Quantitative Real Time – Polymerase Chain Reaction). Each has advantages and disadvantages regarding sensitivity, time to obtain results, and cost. For further information on how to collect samples, ship them, and select assay methods, refer to the instructions on the form.
D. Appel.
July 2009
Is it drought stress or oak wilt?
 Over the past 2-3 months we have been getting quite a number of samples for oak wilt assay. There is a concern that this disease is, once again, becoming a serious epidemic issue. However, the past few years have been difficult for all trees. The extreme drought of 2005 and 2006, and the extreme wet year of 2007 put a tremendous stress on trees. In 2008 and 2009, many parts of Texas still encounter drought situations. All these conditions are stressful to trees. Many different trees have shown declining symptoms, or are showing declining symptoms. Typically stressed trees are more susceptible to invasion by disease pathogens. Is it possible that stressed oaks are predisposed to infection by Ceratocystis fagacearum, the oak wilt fungus? According to Dr. Dave Appel (tree pathologist and oak wilt expert), “NO! There are several tree diseases that stress does not play a factor in infection and oak wilt is one of them. However, wounding does play a role in oak wilt infection.” There is a website online that contains a myriad of good information on oak wilt in Texas.( Link = http://TexasOakWilt.org ). Here you will find out more about oak wilt and some photographs that will help you identify if you might have the problem. This website also provides some advice and options for management of this disease. Oak Wilt confirmation testing takes a minimum of 21-days at the Texas Plant Disease Diagnostic Lab. For submission of samples, please take note of sampling instructions found on page 2 of form D1178.
Over the past 2-3 months we have been getting quite a number of samples for oak wilt assay. There is a concern that this disease is, once again, becoming a serious epidemic issue. However, the past few years have been difficult for all trees. The extreme drought of 2005 and 2006, and the extreme wet year of 2007 put a tremendous stress on trees. In 2008 and 2009, many parts of Texas still encounter drought situations. All these conditions are stressful to trees. Many different trees have shown declining symptoms, or are showing declining symptoms. Typically stressed trees are more susceptible to invasion by disease pathogens. Is it possible that stressed oaks are predisposed to infection by Ceratocystis fagacearum, the oak wilt fungus? According to Dr. Dave Appel (tree pathologist and oak wilt expert), “NO! There are several tree diseases that stress does not play a factor in infection and oak wilt is one of them. However, wounding does play a role in oak wilt infection.” There is a website online that contains a myriad of good information on oak wilt in Texas.( Link = http://TexasOakWilt.org ). Here you will find out more about oak wilt and some photographs that will help you identify if you might have the problem. This website also provides some advice and options for management of this disease. Oak Wilt confirmation testing takes a minimum of 21-days at the Texas Plant Disease Diagnostic Lab. For submission of samples, please take note of sampling instructions found on page 2 of form D1178.
June 2009
My trees are dying, really……
 A tree takes a long time to grow to maturity, yet it seems that it only need a short time for disease to kill it. One of the indications that your tree may be under great duress is the observation of fuzzy gray or olive-green stuff on the trunk or branches of the declining tree. This disease, known as HYPOXYLON CANKER, is caused by the fungus Biscogniauxia atropunctatum (formerly, Hypoxylon atropunctatum). This fungus is believed to be a weak pathogen and may be present on trees. Disease develops when trees are weakened through various stresses, allowing the pathogen to overcome the resistance of the host tree. The last few years has been tremendously stressful on our trees in Texas. The drought in 2005 and 2006, followed by the above-average rains in 2007 has asserted much stress on our trees. Depending on the type of tree, decline or some detrimental effects are observed 1-5 years after the stress incident. This month, we have just posted a series of three factsheets on HYPOXYLON CANKER: 1 – Cause and Biology; 2- Symptoms and signs; 3- Managing the disease.
A tree takes a long time to grow to maturity, yet it seems that it only need a short time for disease to kill it. One of the indications that your tree may be under great duress is the observation of fuzzy gray or olive-green stuff on the trunk or branches of the declining tree. This disease, known as HYPOXYLON CANKER, is caused by the fungus Biscogniauxia atropunctatum (formerly, Hypoxylon atropunctatum). This fungus is believed to be a weak pathogen and may be present on trees. Disease develops when trees are weakened through various stresses, allowing the pathogen to overcome the resistance of the host tree. The last few years has been tremendously stressful on our trees in Texas. The drought in 2005 and 2006, followed by the above-average rains in 2007 has asserted much stress on our trees. Depending on the type of tree, decline or some detrimental effects are observed 1-5 years after the stress incident. This month, we have just posted a series of three factsheets on HYPOXYLON CANKER: 1 – Cause and Biology; 2- Symptoms and signs; 3- Managing the disease.
May 2009
What is that slimy stuff….
 In the past several weeks, I been getting some reports of people wanting to know what is that yellow or white blobs of slime. Sometimes they even leave a slime trail. Splitting them open reveal a spongy, slimy texture much like fluffy meringue. And when they get older, these blobs are hard. When you split these open, they have black sooty centers. These are slime mold. They tend to be more of an aesthetic annoyance than a problem for plants. Slime molds tend to show up around wood, such as wood mulch, in moist and humid conditions. Typically a day or two after heavy rains, or in areas which are well irrigated. Sometimes there is panic when they show up in playgrounds, especially those that uses wood mulch. These myxomycetes are not known to be pathogenic to human. However, some people may be allergic when exposed to the spores (black sooty powder). For more information on slime mold, check out the factsheet. (Click here for factsheet in spanish)
In the past several weeks, I been getting some reports of people wanting to know what is that yellow or white blobs of slime. Sometimes they even leave a slime trail. Splitting them open reveal a spongy, slimy texture much like fluffy meringue. And when they get older, these blobs are hard. When you split these open, they have black sooty centers. These are slime mold. They tend to be more of an aesthetic annoyance than a problem for plants. Slime molds tend to show up around wood, such as wood mulch, in moist and humid conditions. Typically a day or two after heavy rains, or in areas which are well irrigated. Sometimes there is panic when they show up in playgrounds, especially those that uses wood mulch. These myxomycetes are not known to be pathogenic to human. However, some people may be allergic when exposed to the spores (black sooty powder). For more information on slime mold, check out the factsheet. (Click here for factsheet in spanish)
March 2009
Outbreak of rust on cedars
 Recently we have received reports of ‘yellow goo’ on cedar trees from east Texas to the Dallas –Fort Worth areas. These ‘yellow goo’ are telia structure of the rust fungi. It is in the telia that teliospores develop and are released to cause new infections on the alternate host, typically rosaceous plants such as apples, pears, quinces, hawthorns, and many others. The question that we get asked most often is ‘how do we treat the problem (on the cedar)?’ The simple answer is nothing right now. This is because the spores (teliaspores) that are release do not infect the cedar but instead infects the alternate host. The fungal agents causing this disease problem have a distinct 2-host cycle, meaning it must spend part of its life on both host. Preventative treatments on the susceptible rosaceous hosts now will help to suppress potential rust infections. There are 2 common rusts that occur in Texas: cedar-apple rust and cedar-quince rust. These two diseases can be differentiated based on the galls characteristics on cedars. Cedar-apple rust, caused by the fungus Gymnosporangium juniperi-virginianae, usually manifest itself as hard greenish to brown spherical galls. Whereas cedar-quince rust, caused by Gymnosporangium claviceps, causes long elongated swelling on twigs and branches of the cedar. When the telia develops, cedar-apple rust then to look like little round yellow balls and cedar-quince looks more like a flat slime goop on the branches. More information (factsheets) are available to describe these disease problem and can be downloaded from this website: cedar-apple rust and cedar-quince rust. A simple approach to managing this disease is to remove the galls on the cedar. This may potential reduce the inoculum that may be present to cause new rust infections. However, it should be noted that it is easier to identify and remove cedar-apple galls than recognizing cedar-quince galls.
Recently we have received reports of ‘yellow goo’ on cedar trees from east Texas to the Dallas –Fort Worth areas. These ‘yellow goo’ are telia structure of the rust fungi. It is in the telia that teliospores develop and are released to cause new infections on the alternate host, typically rosaceous plants such as apples, pears, quinces, hawthorns, and many others. The question that we get asked most often is ‘how do we treat the problem (on the cedar)?’ The simple answer is nothing right now. This is because the spores (teliaspores) that are release do not infect the cedar but instead infects the alternate host. The fungal agents causing this disease problem have a distinct 2-host cycle, meaning it must spend part of its life on both host. Preventative treatments on the susceptible rosaceous hosts now will help to suppress potential rust infections. There are 2 common rusts that occur in Texas: cedar-apple rust and cedar-quince rust. These two diseases can be differentiated based on the galls characteristics on cedars. Cedar-apple rust, caused by the fungus Gymnosporangium juniperi-virginianae, usually manifest itself as hard greenish to brown spherical galls. Whereas cedar-quince rust, caused by Gymnosporangium claviceps, causes long elongated swelling on twigs and branches of the cedar. When the telia develops, cedar-apple rust then to look like little round yellow balls and cedar-quince looks more like a flat slime goop on the branches. More information (factsheets) are available to describe these disease problem and can be downloaded from this website: cedar-apple rust and cedar-quince rust. A simple approach to managing this disease is to remove the galls on the cedar. This may potential reduce the inoculum that may be present to cause new rust infections. However, it should be noted that it is easier to identify and remove cedar-apple galls than recognizing cedar-quince galls.
February 2009
Turf problem SPRINGing up?
 In this state of Texas, we grow many different turfgrasses for our enjoyment: bermudagrass, zoysia, St. Augustine grass, buffalograss and others. With spring just round the corner, there is great anticipation for the return of nice, lush, dense green lawn and the smell of freshly cut grass. But this ideal situation does not occur for everyone. Many times there are areas of grass thinning, and even bare spots. Common grasses found on landscapes are St. Augustine grass and bermudagrass. A disease that is common to both grasses is one that is caused by a fungus, Gaumannomyces sp.. The common name of this disease is “take-all patch” on St. Augustine, and is known as “bermudagrass decline” on bermudagrass. The symptoms of this disease are typically yellowish thinned out large irregular patches. Closer inspection of the grass may show discolored stolons (runners) and a poor root system (black, short and dry rotted). Additional information about this disease can be found on the AgriLife Extension factsheet “Take-all root rot of turfgrass” (L-5170) *.
In this state of Texas, we grow many different turfgrasses for our enjoyment: bermudagrass, zoysia, St. Augustine grass, buffalograss and others. With spring just round the corner, there is great anticipation for the return of nice, lush, dense green lawn and the smell of freshly cut grass. But this ideal situation does not occur for everyone. Many times there are areas of grass thinning, and even bare spots. Common grasses found on landscapes are St. Augustine grass and bermudagrass. A disease that is common to both grasses is one that is caused by a fungus, Gaumannomyces sp.. The common name of this disease is “take-all patch” on St. Augustine, and is known as “bermudagrass decline” on bermudagrass. The symptoms of this disease are typically yellowish thinned out large irregular patches. Closer inspection of the grass may show discolored stolons (runners) and a poor root system (black, short and dry rotted). Additional information about this disease can be found on the AgriLife Extension factsheet “Take-all root rot of turfgrass” (L-5170) *.
* can be downloaded at http://AgriLifebookstore.org
January 2009
New in 2009
 As this year begins, there are several changes that are being and have been implemented at the Texas Plant Disease Diagnostic Laboratory. Our lab is currently undergoing minor renovations to create better workflow and allow us to more efficiently receive and handle plant disease samples. Signage is being upgraded for better visibility to visitors. Most importantly, there are new forms for the submission of samples. These forms can be found on this website under FORMS. Two new forms are available: (1) General Diagnostic Form D-1178 – is used for all general submission for diagnostics, (2) Nematode Detection Assay Form D-827 – used specifically for nematode analysis. These new forms were designed to help up gather necessary information for accurate diagnosis, and even expand our services to homeowners on gardening through our Texas AgriLife Extension Service County offices when additional assistance is needed. For contact information to your local AgriLife Extension County office, please check the directory at http://AgriLifeExtension.tamu.edu . General gardening questions can typically be answered at the local offices.
As this year begins, there are several changes that are being and have been implemented at the Texas Plant Disease Diagnostic Laboratory. Our lab is currently undergoing minor renovations to create better workflow and allow us to more efficiently receive and handle plant disease samples. Signage is being upgraded for better visibility to visitors. Most importantly, there are new forms for the submission of samples. These forms can be found on this website under FORMS. Two new forms are available: (1) General Diagnostic Form D-1178 – is used for all general submission for diagnostics, (2) Nematode Detection Assay Form D-827 – used specifically for nematode analysis. These new forms were designed to help up gather necessary information for accurate diagnosis, and even expand our services to homeowners on gardening through our Texas AgriLife Extension Service County offices when additional assistance is needed. For contact information to your local AgriLife Extension County office, please check the directory at http://AgriLifeExtension.tamu.edu . General gardening questions can typically be answered at the local offices.
The Texas Plant Clinic will also be introducing several new and/or enhanced services aimed at our commercial growers through collaboration with Plant Pathology specialists. Stay tuned for more news.
November 2008
 Recently, the Plant Disease Clinic got in an email from one of our County AgriLife Extension horticulture agents requesting help with identifying some “odd thing” that was observed on hickory leaves. This “odd thing” was described as white crystals on the underside of the leaves. From the photos, both the plant pathologist and entomologists thought that it might be a result of a stress response to insect feeding. When we received the physical sample at TPDDL, we took a closer look at it. And those crystals looks awfully like spores. We did confirm that it was fungal spores and then we began to look at what type of fungus it might be. Through some research, we found an old publication that described the problem on pecans. Apparently, this problem occurs on pecan and is commonly known as “leaf mold”. The scientific name of this fungus is Articularia quercina. It is a problem when environmental conditions are conducive to the disease development: post- rainy conditions, high humidity and the tree is of poor vigor. A journal article that described details of this fungus was published by Vera Charles in Mycologia in 1935. Click here for a link to the paper published by the Mycological Society of America. Treatment for this problem is not warranted at this time of the year as the tree will begin to lose its leaves in preparation for the winter. One can practice sanitation by removing the fallen leaves and destroying them to reduce the amount of inoculum (spore that might cause new infections) for the following season.
Recently, the Plant Disease Clinic got in an email from one of our County AgriLife Extension horticulture agents requesting help with identifying some “odd thing” that was observed on hickory leaves. This “odd thing” was described as white crystals on the underside of the leaves. From the photos, both the plant pathologist and entomologists thought that it might be a result of a stress response to insect feeding. When we received the physical sample at TPDDL, we took a closer look at it. And those crystals looks awfully like spores. We did confirm that it was fungal spores and then we began to look at what type of fungus it might be. Through some research, we found an old publication that described the problem on pecans. Apparently, this problem occurs on pecan and is commonly known as “leaf mold”. The scientific name of this fungus is Articularia quercina. It is a problem when environmental conditions are conducive to the disease development: post- rainy conditions, high humidity and the tree is of poor vigor. A journal article that described details of this fungus was published by Vera Charles in Mycologia in 1935. Click here for a link to the paper published by the Mycological Society of America. Treatment for this problem is not warranted at this time of the year as the tree will begin to lose its leaves in preparation for the winter. One can practice sanitation by removing the fallen leaves and destroying them to reduce the amount of inoculum (spore that might cause new infections) for the following season.
October 2008
Poinsettia Headaches
 In the past few weeks, we have been seeing a lot of poinsettias being sent in for problem diagnosis. Typical complaints were that the plants were wilting and not recovering. Usually we look for damage on stem and on roots because wilting is a symptom that indicates that that plant is not getting water to those leaves. Some usual suspects would include dried and/or rotted roots. These symptoms can be caused by too much or too little water.
In the past few weeks, we have been seeing a lot of poinsettias being sent in for problem diagnosis. Typical complaints were that the plants were wilting and not recovering. Usually we look for damage on stem and on roots because wilting is a symptom that indicates that that plant is not getting water to those leaves. Some usual suspects would include dried and/or rotted roots. These symptoms can be caused by too much or too little water.
And when a plant is stressed, it is more susceptible to attacks by pathogens. Some common root and/or crown rot fungal pathogens are Thielaviopsis (common name = black root rot), Phytophthora , Pythium and Fusarium. If we find some indications that these fungi are present, we typically try to isolate it (i.e grow it out on selective media in petri dishes) for confirmation. This procedure take several days.
Some things that you can do to prevent these sorts of fungal problems are to insure that your plants are not excessively stressed. This would include making sure that you are irrigating properly, no overwatering or fertilizing. If there is a history with a particular pathogenic problem, you may want to implement a preventative management program.
For additional help or information, you can contact your local county AgriLife Extension office.
Changing of the guard at the TPDDL – New director at TPDDL
The Urban Plant Pathology program is currently taking a little break. Dr. Kevin Ong has been reassigned to be the Director of the Texas Plant Disease Diagnostic Laboratory in College Station. He assumes this position on September 1, 2008. Programs such as the Education Community Gardens at Dallas will continue under the guidance of Dr. Fouad Jaber and Dr. John Sloan. Third Saturday programs will continue through this year.
The Texas Plant Disease Diagnostic Laboratory is based in College Station and provide plant disease diagnostic support to AgriLife Extension personnel, various plant industries and the general public. This fee-based service strived to provide quality diagnostic support in a timely fashion.
Spring 2008
Challenges in starting seeds at home
A major challenge to the home-gardener starting seedlings indoors is creating the proper environment for the seeds to germinate. One problem is maintaining proper soil moisture; however, it is easy to overdo this. Seeds do need soils that are moist, but not wet! Wet soils cause stress to the seedlings and also create an environment where several seedling pathogens (disease-causing agents) can thrive. “Damping-off” is the common name of a disease typically found on seedlings and exacerbated by an overly wet environment. The causative agent in this case is a fungus; Pythium (Pee-tee-um) and Phytopthtora (Phi-tow-for-rah) are two common fungi that cause this ailment. These fungi love water and wet soils making a damping-off problem all the worse. Damping-off
March 2008 – Something wrong with live oaks?
My oak tree is dying! Help! “ This is a common cry from some homeowners in the Dallas-Fort Worth metroplex in the months of March and April. The Texas live oak is a beautiful stately tree that retains its leaves in the winter, hence also known as the evergreen oak. However, as spring arrives, this tree starts to look sickly. Leaves turn yellow. In some cases, there can be brown or black spots on it. Soon after, these leaves start to drop. When the leaves drop, some branches look bare. But HAVE NO FEAR. This is NORMAL. This is the time when these trees begin to put out new leaves. In order for the new leaves to come in, the old had to be discarded. So look carefully to make sure that you needlessly panic for a natural occurrence.
Check out this fact sheet for more detailed information. LiveOak


